As more patients with cancer have their tumors genomically profiled, and more therapies targeting genomic alterations enter clinical trials, the task of connecting patients to trials for which they are eligible can be especially challenging. A computer platform developed at Dana-Farber Cancer Institute makes the matching process both easier and faster, its designers report in a new study.
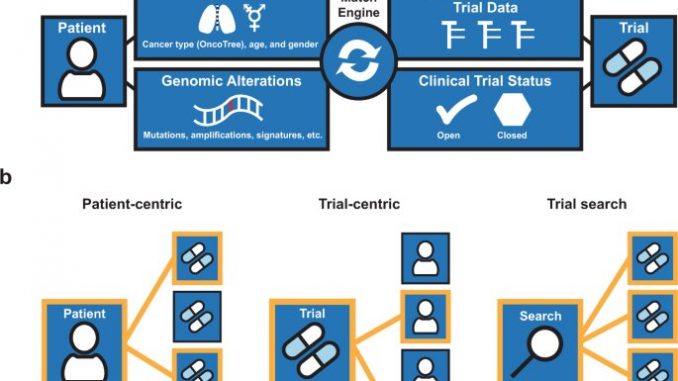
Called MatchMiner, the platform helps clinicians and clinical researchers find potential matches between patients and targeted therapy trials based on genetic alterations in patients’ tumors. During a five-year period at Dana-Farber, it helped bring about roughly one in every five consents to join precision medicine trials among patients with genomic data in MatchMiner. It also sped the process of enrolling patients in such trials by more than 20%, found the authors of the study, published in npj Precision Oncology.
“Profiling patient tumors for genomic alterations has become a widespread part of cancer care, especially as new drugs targeting those alterations go into clinical trials or are approved as cancer therapies,” says Tali Mazor, Ph.D., the co-lead author of the paper with Dana-Farber colleague Harry Klein, Ph.D. “The combination of this growing body of genomic data and increasing number of precision medicine trials has created a kind of disconnect: finding the right trial for each patient can be a difficult task. MatchMiner helps bridge that gap.”
The platform, developed by the Knowledge Systems Group at Dana-Farber led by Ethan Cerami, Ph.D., and Michael Hassett, MD, MPH, draws on Dana-Farber’s extensive programs in genomic analysis and clinical research. Over the past decade, more than 40,000 patients at the Institute have had their tumor tissue analyzed for alterations in over 400 cancer-related genes. The Institute and its partners lead or participate in thousands of clinical trials, including about 450 involving targeted therapies since 2017. MatchMiner, which was launched in 2016, links these systems to help match patients to appropriate trials.
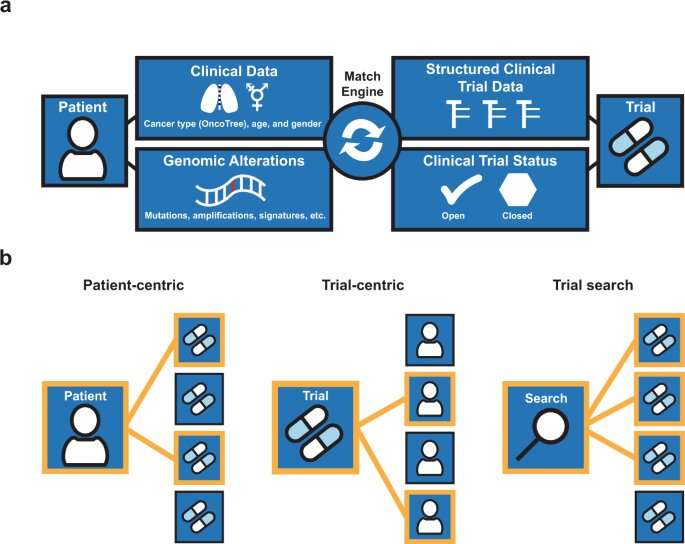
“MatchMiner can be used by an oncologist or other clinician to look up trial options for an individual patient,” Klein remarks. “Or it can be used by a trial team to identify potential trial participants by setting up a genomic filter that screens candidates for specific genomic criteria.”
Unlike most other matching platforms, which are designed for use at a single cancer center, MatchMiner is open-source and can be adapted by other institutions. As new trials open at Dana-Farber, a curator reviews them to see if they should be included in MatchMiner, ensuring the platform is up-to-date. As of March 2021, 354 precision medicine trials are integrated into MatchMiner.
In the new paper, investigators analyzed enrollment data for precision medicine trials at Dana-Farber to determine whether MatchMiner expedited the process of finding an appropriate trial for patients whose tumors had been genomically profiled. The researchers found 166 instances in which the platform identified a potential match between a patient and a trial, and the trial team or the patient’s oncologist viewed the match, leading to the patient’s consent to join the trial.
To further assess the impact of the platform, investigators compared the “time to consent”—the time between the genetic profiling of a tumor and the patient’s consent to participate in the trial—for the 166 consents obtained via MatchMiner and for 353 consents obtained without the platform. “We found the time to consent for the MatchMiner group was 55 days faster than for the non-MatchMiner group, an improvement of 22%,” Klein says.
MatchMiner links patients to trials not only by the molecular features of the patient’s tumor but also by patient age and tumor type. Other trial criteria, such as tumor stage, previous treatment, and patient’s overall health are not considered for a patient-trial match. As a result, the matches proposed by MatchMiner are preliminary and need to be followed up to ensure that patients meet all the trial criteria, researchers say.
Source: Read Full Article