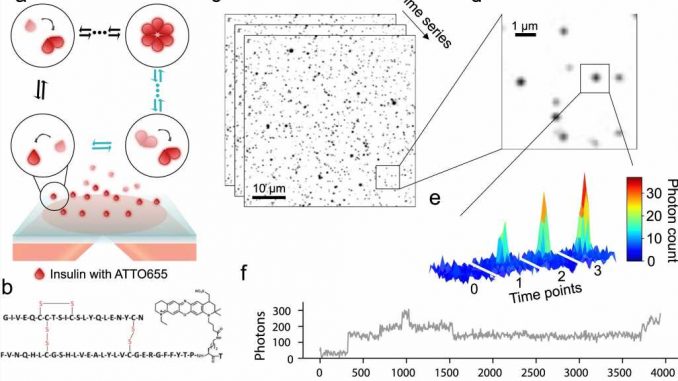
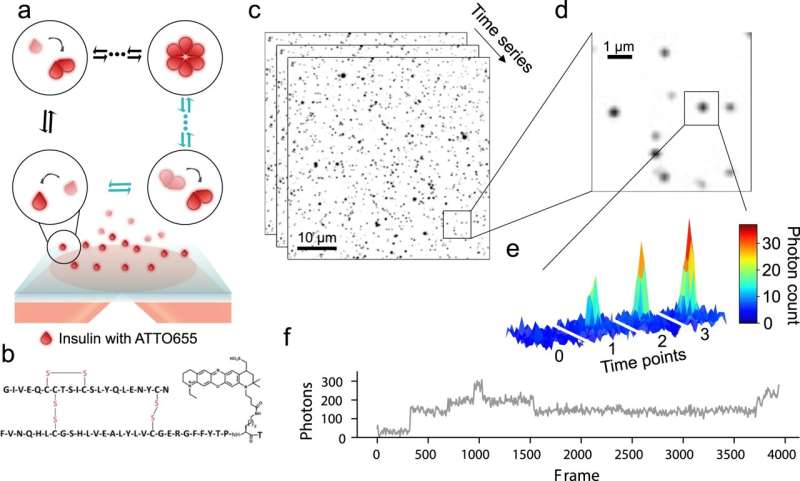
New research suggests that a large portion of a diabetic’s insulin dose is unlikely to work as expected. University of Copenhagen researchers have discovered that we have miscalculated insulin behavior for many years. The discovery provides a tool for developing better insulin preparations that are depended upon by millions of people worldwide.
If you are one of the many millions of type 1 diabetics worldwide, you know that there is a difference in how rapidly and for how long insulin preparations work in the body. For diabetics, these differences are crucial for effective treatment. Getting too little or too much insulin can lead to blood sugar that is either too low or high. Both conditions can be dangerous.
The absorption of insulin in the body is controlled by how insulin molecules assemble themselves in clusters. Whereas a single molecule provides rapid action in the body, clusters of six molecules—known as hexamers—are long-acting. For decades, it has been assumed that insulin assembles with a certain distribution of molecular clusters of either one, two or six molecules. Pharmaceuticals have been designed based upon this assumption.
But with the help of highly advanced single molecule microscopy, researchers at the University of Copenhagen, in collaboration with Aarhus University, have become the first to demonstrate that this important point has been wrong for years.
“It is now apparent to us that we’ve gotten things wrong by 200 percent. There are only half as many single molecules in insulin compared to what we thought. Conversely, there are far more six-molecule clusters than we assumed. Ultimately, if this is basic research insights apply in human body, it means that when we believe to be administering a certain dose, it may only be half the dose with the rapid-acting effect in the body that we had expected,” says Professor Nikos Hatzakis of the Department of Chemistry, the study’s lead author.
In other words, much of the insulin that diabetics currently put into their bodies may not actually be getting absorbed as expected. While the researchers emphasize that this is not outright dangerous for patients, it does show that there is great potential for the development of more precise medications.
The study, Enhanced Hexamerization of Insulin via Assembly Pathway Rerouting Revealed by Single Particle Studies, has just been published in Communications Biology.
From a crude model to detailed view
“Insulin preparations have only gotten better and better over the years, and a great many diabetics are well regulated. However, the development of insulin preparations has been based on a certain assumption about how the molecules assemble. With the crude standard model, this process was never been appreciated at a detailed level. That’s what we can do,” says the study’s other lead author, Professor Knud J. Jensen, of the Department of Chemistry.
“This doesn’t mean that current insulin medications are bad or that patients have been medicated wrongly. But we now have a basic understanding of how insulin behaves and how much could be available to the body as rapid-acting medication. We now have the right method for providing us with accurate figures. We hope that the industry will use this or a similar tool—both to check current insulin preparations and to develop new ones,” adds Nikos Hatzakis.
The research results were achieved through a mix of chemistry, machine learning, simulations and advanced microscopy. The Department of Chemistry researchers began by directly observing the process in which each insulin molecule joins forces with other molecules to assemble into clusters. This allowed them to see how fast each cluster forms. The researchers looked at about 50,000 clusters.
Knowing the exact distribution of different clusters in a given amount of insulin is fundamental when developing medications that need to have either short- or long-acting effects in the body:
“The clustering of insulin is incredibly important for how preparations work. Because the difference between a rapid- and slow-acting insulin preparation is dependent upon how quickly the molecules assemble in clusters and how quickly they disassemble. Access to highly advanced equipment makes it relatively simple and fast to gain insight into exact concentrations, knowledge that at the same time, is also quite sophisticated,” says lead author Freja Bohr, a Ph.D. fellow in Nikos Hatzakis’ research group at the Department of Chemistry.
Better insulin benefits millions
In addition to the different distribution of molecular clusters, the observations also show that cluster formation is a much more complex process than once presumed. The clusters can both grow and shrink at far more different intervals than previously supposed.
“Without being able to say exactly how just yet, this should make it possible to expand the number of ways in which preparations are designed. This could lead to an insulin with a different effect profile that reduces the fluctuations in patients’ blood sugar—which remains a major challenge,” says Freja Bohr.
Knud J. Jensen, who has been researching insulin for more than 15 years, believes that the new knowledge will be able to optimize all types of new insulin and make a difference for the more than 40 million children and adults who take insulin on a daily basis. Life as a diabetic is still not without trouble:
“I sometimes receive inquiries from parents who ask if there is something better for treating their young children. When a person has poorly regulated type 1 diabetes, they can feel awful for long periods of time. Among other things, they can wake up with nightmares, feel unwell due to low or high blood sugar concentrations, risk losing consciousness due to low blood sugar and suffer consequential damage to their eyes and feet later on in life. So, if life can be made better for children by making better insulin, that’s fantastic,” says Jensen.
More information:
Freja Bohr et al, Enhanced hexamerization of insulin via assembly pathway rerouting revealed by single particle studies, Communications Biology (2023). DOI: 10.1038/s42003-022-04386-6
Journal information:
Communications Biology
Source: Read Full Article